Mon Feb 10 08:26:08 UTC 2025: **Indian Company Receives License for Groundbreaking Lumpy Skin Disease Vaccine**
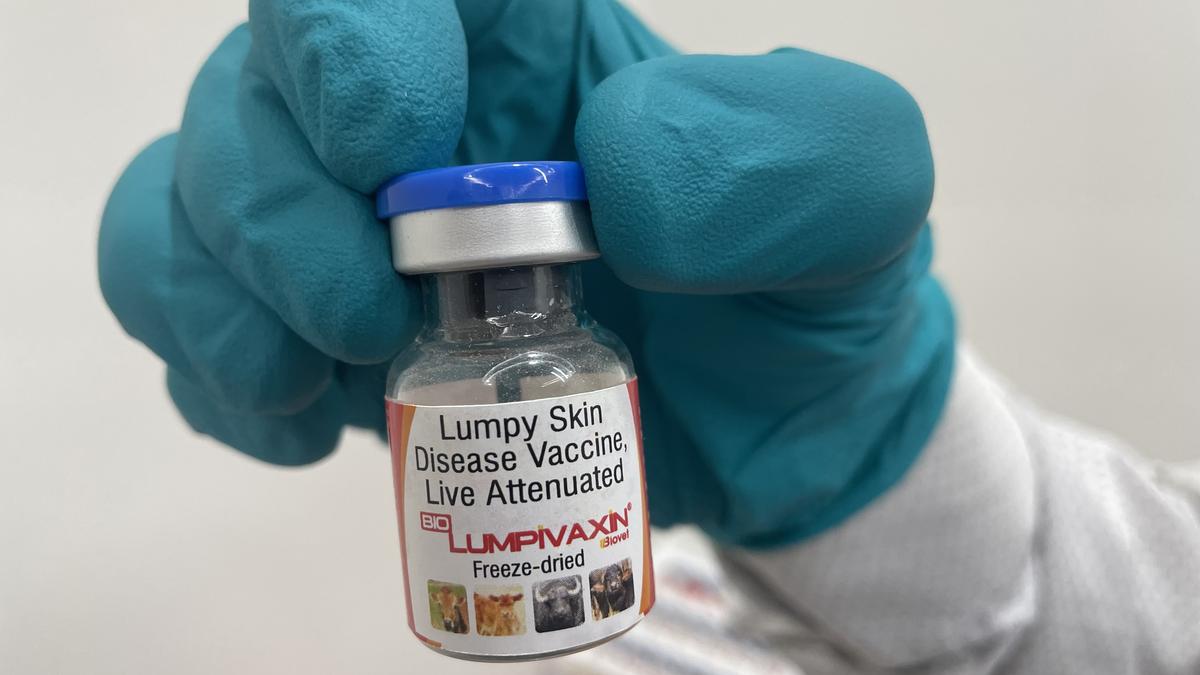
HYDERABAD, India (February 10, 2025) – Biovet, an animal health vaccine maker and a subsidiary of Bharat Biotech, announced today that it has received Central Drug Standards Control Organization (CDSCO) approval for its lumpy skin disease (LSD) vaccine, Biolumpivaxin. This vaccine is the first globally to utilize a DIVA (Differentiation of Infected from Vaccinated Animals) marker, allowing for clear distinction between vaccinated and naturally infected animals.
Developed in collaboration with the Indian Council of Agriculture Research (ICAR), Biolumpivaxin boasts a high safety and efficacy profile. The freeze-dried vaccine requires a single annual dose for cattle and buffaloes over three months old. Biovet’s facility in Mallur, Karnataka, has the capacity to produce 500 million doses annually.
The vaccine, a result of three years of research, is a significant advancement in combating LSD, a disease that has devastated India’s cattle population in recent years, resulting in an estimated 200,000 deaths and significant milk production losses. The novel vaccine was developed using a virus strain from ICAR-National Research Centre on Equines, Hisar, and rigorously tested at ICAR-NRCE and the Indian Veterinary Research Institute (IVRI).
Biovet founder Krishna Ella hailed the CDSCO approval as a major step towards India’s self-reliance in veterinary healthcare, eliminating dependence on imported vaccines. He emphasized the vaccine’s crucial role in disease surveillance, eradication programs, and the sustainability of the dairy industry. The vaccine will be commercially available shortly.
