Sat Oct 11 00:05:00 UTC 2025: **News Article:**
**Cough Syrup Tragedy Claims Lives of 24 Children in Madhya Pradesh**
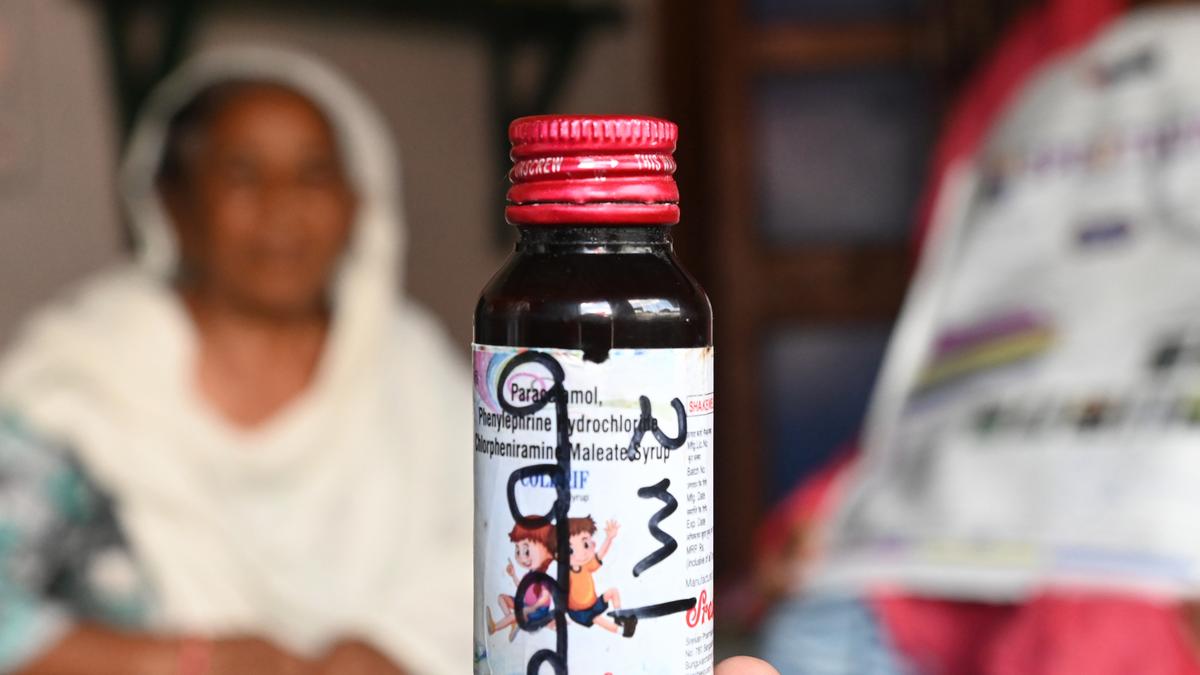
**Parasia, Madhya Pradesh, October 11, 2025** – A deadly cough syrup contamination has resulted in the tragic deaths of 24 children in Madhya Pradesh since September. The culprit is believed to be Coldrif cough syrup, manufactured by Tamil Nadu-based Sresan Pharmaceuticals. The children, mostly from the Chhindwara district, developed severe kidney damage after consuming the syrup, which was prescribed by local doctors for fever and cold symptoms.
Symptoms included stomach aches, vomiting, drowsiness, and swelling, ultimately leading to kidney failure. Distraught parents recount the rapid deterioration of their children’s health after administering the medication.
Investigations by state and central health authorities revealed the presence of diethylene glycol (DEG), a toxic chemical, in the cough syrup. DEG is a cheaper alternative to pharmaceutical-grade solvents and has been found to replace them for its similar taste and consistency.
Sresan Pharmaceuticals’ manufacturing license has been suspended, and the owner, G. Ranganathan, has been arrested. A local doctor who prescribed the syrup has also been arrested, raising concerns about the potential for such contaminations not being easily detected without testing procedures.
The incident has exposed serious flaws in India’s drug regulatory system. While the Central Drugs Standard Control Organization (CDSCO) approves new drugs, manufacturing licenses are handled at the state level, leading to potential gaps in quality control. Inspections, meant to be conducted regularly, have been lax, with some facilities in Tamil Nadu not being inspected for six years.
Health officials are now scrambling to locate and seize remaining bottles of the contaminated syrup. The CDSCO has ordered a joint audit of cough syrup manufacturers across the country.
This tragedy echoes similar incidents in recent years, including the deaths of over 300 children in Gambia, Indonesia, and Uzbekistan in 2022 due to contaminated Indian-made cough syrups, raising serious questions about the safety and regulation of pharmaceutical products in India.
